In the world of high-precision biology, a peptide is only as good as its conformational stability. Because peptides are essentially “biological messages,” any break in their amino acid chain renders the message unreadable to cellular receptors. Understanding the science of half-life and storage is not just about avoiding “expired” products—it is about ensuring experimental reproducibility.
This technical article explores the factors that govern peptide degradation and provides a standardized data sheet for maximizing the lifespan of your research materials.
The Two Faces of Half-Life
When researchers discuss “half-life,” they are often referring to two very different metrics. Distinguishing between them is critical for accurate data collection:
- 1. Storage (Chemical) Half-Life: This refers to how long the peptide remains stable and intact within its vial (lyophilized or liquid) before oxidation or hydrolysis begins to break the peptide bonds.
- 2. Biological (In Vivo) Half-Life: This refers to the time it takes for a peptide to be cleared from a research model’s system after administration. Many natural peptides have a biological half-life of only minutes, while modified synthetic versions are engineered to last hours or days.
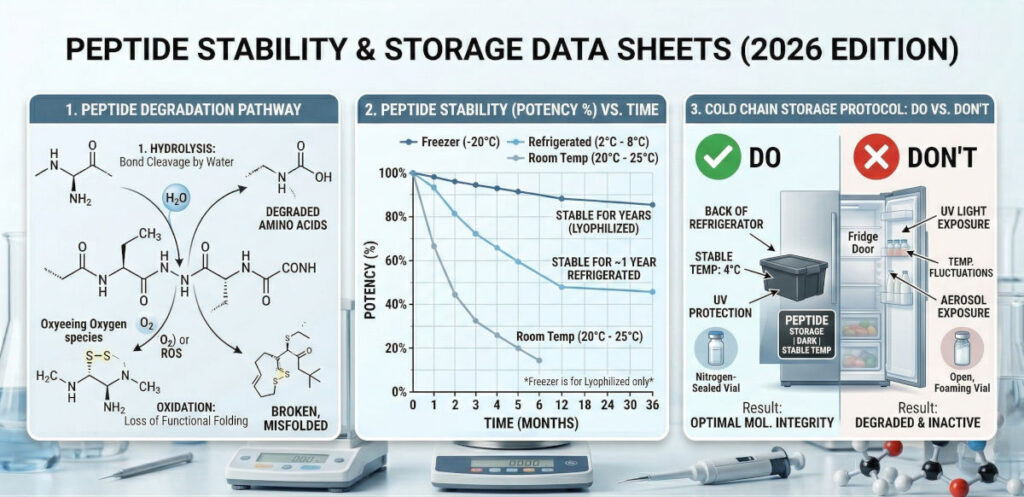
The Three Enemies of Peptide Stability
Peptide degradation is a chemical reaction. To slow this reaction, researchers must mitigate the “Three Enemies” of molecular integrity:
1. Temperature (Kinetic Energy)
Heat increases molecular vibration. At high temperatures, the peptide bonds can snap, or the peptide may “unfold” (denature). While some sequences are thermostable, most regenerative peptides begin to degrade rapidly if left at room temperature for extended periods.
2. Light (Photolysis)
UV radiation provides enough energy to break chemical bonds directly. This process, known as photolysis, can turn a high-purity clear solution into an inactive, degraded mixture within hours of direct sunlight exposure.
3. Moisture (Hydrolysis)
Water is the medium of life, but it is also the medium of decay. In a process called hydrolysis, water molecules can react with peptide bonds, effectively “cutting” the Lego chain back into individual, useless amino acids.
Stability Data Sheet: Storage Temperature Protocols
To ensure the highest quality results, follow these standardized storage timelines based on the physical state of the peptide:
| Storage Condition | Lyophilized (Powder) | Reconstituted (Liquid) |
|---|---|---|
| Room Temp (20°C – 25°C) | Stable for 2–4 weeks | Degrades in 24–48 hours |
| Refrigerated (2°C – 8°C) | Stable for 12–24 months | Stable for 4 weeks |
| Freezer (-20°C) | Stable for 3–5 years | NOT RECOMMENDED* |
*Freezing reconstituted peptides can cause ice crystals to form, which may shear the delicate molecular chains unless specialized cryoprotectants are used.
Advanced Handling: The “Aliquoting” Protocol
One of the most common causes of peptide degradation in the lab is Freeze-Thaw Cycling. If you store a 10mg vial in the freezer and take it out every day to draw a dose, the constant change in temperature will destroy the sequence faster than simple refrigeration.
The Aliquoting Protocol is the professional solution: If you have a large amount of lyophilized powder, divide it into smaller, single-use vials while dry. Only reconstitute what you intend to use within a 30-day window. This minimizes exposure to both oxygen and temperature fluctuations.
Oxidation and Secondary Structure
Some peptides contain specific amino acids, like Cysteine or Methionine, which are highly susceptible to oxidation. When these amino acids “rust,” they can form incorrect disulfide bridges, causing the peptide to fold into the wrong shape. Once a peptide loses its secondary structure, it can no longer dock with its receptor, rendering it biologically inert.
Technical Stability FAQ
In most cases, if the peptide was in its lyophilized (powder) form, it will be perfectly fine. However, if it was already reconstituted and left at room temperature for over 12 hours, its potency has likely begun to drop. It is not “toxic,” but its experimental accuracy is compromised.
High-quality manufacturers seal peptide vials under a vacuum or inert gas (like Nitrogen) to remove oxygen. This is a primary defense against oxidation. If you hear a small “hiss” of air when you first insert a needle, it is a sign that the seal was intact and the peptide was protected.
The “puck” is the result of lyophilization (freeze-drying). A solid, intact puck is usually a sign of a high-quality, moisture-free environment. If the powder looks melted or sticky (deliquescence), it may have been exposed to moisture and should be used with caution.
Summary
Peptide research is an investment in data. Protecting that investment requires a strict adherence to cold-chain protocols. By maintaining a stable temperature, protecting from UV light, and utilizing proper reconstitution math, you ensure that every microgram of your peptide delivers the precise biological signal your research demands.
For more technical data and high-purity sequences, view our full research collection.
